Russian export of syringe:

Manufacturers of Russian Syringe wholesale:
Russian Syringe wholesale products

Syringe SHRG-250, syringe SHRG-630 of our own production on
- 0
- 2525
- 2

Sausage syringe horizontal biowin 5 kg
- 1
- 3070
- 3

Sausage syringe horizontal EdelHoff 2, 5 kg
- 1
- 2894
- 2

Sausage syringe vertical biowin 3 kg
- 2
- 2547
- 1

Sausage syringe 4 kg oscar cook
- 0
- 3020
- 1

Injector for injection PSS VNU 159 vacuum filler
- 0
- 1733
- 0

Vacuum screw syringe Vemag Robot 500
- 2
- 1830
- 5

Vacuum rotary and screw syringes sale
- 1
- 1253
- 4
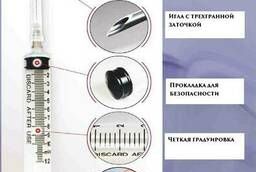
Syringe: 1, 2-2. 5, 3, 5, 10, 20, 50 ml. Wholesale. (Bu Kwang, Korea)
- 0
- 1113
- 4

Screw Pile Blades without Taper - Syringe
- 0
- 1167
- 1

Electric gear syringe dispenser COMBI (Italy)
- 0
- 1285
- 0

Dispenser-syringe for fillings with attachments for decorating DSH-37
- 2
- 2350
- 0

Horizontal Sausage Syringe HS-7L Foodatlas
- 0
- 1424
- 3

Horizontal sausage syringe TG-7L
- 2
- 1134
- 0

Vacuum sausage syringes for sale
- 2
- 1457
- 2

Sausage vacuum filler vemag robot 700
- 1
- 1491
- 5

Syringe injector for impregnation of meat
- 0
- 1256
- 2

Confectionery dispenser-syringe 7
- 1
- 1147
- 2
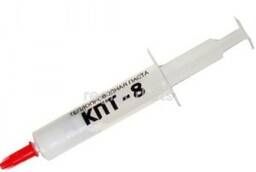
Heat-conducting paste KPT-8 2ml (syringe) ... 09-3751
- 0
- 1455
- 2

Vacuum rotary syringe GZY 2000
- 1
- 1184
- 5
Medical disposable syringes
- 0
- 4709
- 0

Syringe ShRG-630
- 0
- 1069
- 4

Screw lubricating gun ShVS
- 0
- 576
- 5

Syringe SHRG-250 from the manufacturer (Russia)
- 2
- 1027
- 3

Automatic syringe dispenser Leader- 2 m
- 0
- 1425
- 0

Nozzle for a syringe pump lubricant 4-lobe
- 1
- 1891
- 0

Sausage syringe nozzle shv-1
- 1
- 1135
- 4

Hamburger Sausage Nozzle
- 2
- 819
- 0

Janets syringe 3-component ELETS, 150 ml. Set 72. ..
- 1
- 1130
- 4

Pneumatic syringe DG-Q02
- 2
- 1119
- 2
🇷🇺 TOP Exporters Syringe from Russian Federation
Syringe manufacturing companies you many buy this products:
Single-use syringe according to TVNL.942311.025 TU, execution options: 1. Single-use syringe, three-piece, with a catheter tip with a nominal capacity of 50 ml. (type 260600). 2. Disposable syringe
Set of instruments for urological operations according to TU 9437-012-10625445-2006: Nozzle valve; Syringe-evacuator; Syringe adapter for rigid connection; Syringe adapter for flexible connection; Barrel with obturator 1
Single-use injection syringe, sterile 50 ml according to TU 32.50.13-005-11701993-2017 in the following versions: 1. Syringe 50 ml Luer type with or without a needle. 2. Syringe 50 ml Luer-Lock type with or without a
Technological equipment for the food industry: gear syringe dispenser model DSH-37, piston syringe dispenser model DP-37, machine for filling filling into wafer products model DM Н-2
TempeLight temporary filling material of light curing according to TU 9391-019-40151387-2007 Completeness: - syringe with white TempeLight material - 1 pc. - syringe with blue TempeLight material - 1 piece
Endoprosthesis of synovial fluid REVISK (REVISK) in syringes according to TU 9398-002-01370391-2016: consisting of: 1. Filled sterile syringe 2.0 ml - 1 pc. 2. Instructions for use - 1 pc. 3. Sticker-insert with batch nu
Oxaliplatin-TEVA, concentrate for solution for infusion 5 mg ml, 10 ml, bottle (1), cardboard pack, [adapter to bottle + adapter to syringe + adapter for syringe injection] box, RU LSR-00540309 from 06.07.2009 (date p
Gel for etching enamel and dentin Celite Prodent 36 according to TU 9391-017-10614163-2011 in the following configuration: - Syringe with 5 ml gel (weight 7.5 g) closed with a cap; - Nozzle for a syringe (needle) in
Oxaliplatin-TEVA, concentrate for solution for infusion 5 mg ml, 10 ml, bottle (1), cardboard pack, [adapter to bottle + adapter to syringe + adapter for syringe injection] box, RU LSR-00540309 from 06.07.2009 (date p
Syringe for single use injection, sterile 50 ml according to TU 9398-005-11701993-2014 consisting of: 1. Syringe, complete (1 pc.) ... 2. Injection needle (1 pc.). 3. Consumer container (1 pc.).
Technological equipment for the food, meat and dairy industry: Dosing and forming equipment, Vacuum syringe
Syringes-fillers brands HAKKA, GEMLUX, GASTRORAG, models: GL-SV -3, GL-SV-5, GL-SV-7, GL-SV-10, GL-SV-12, GL-SV-3SS, GL-SV-5SS, SH-3, SH-5, SH-7 , SV-3, SV-5, SV-7
Processed beekeeping products for the food industry: beekeeping, royal jelly, lane ha, pollen, propolis, brand LipkoSladko LipkoSladko, packed in plastic, glass, cans, capsules, syringes
Confectionery flour products: a tartlet for snacks, a syringe with cottage cheese, a cottage cheese cake, a shortbread cake with cottage cheese, a rum baba, a white biscuit basic semi-finished product, biscuit with cocoa
Technological equipment for the food industry: jigging machines, syringe-dispensers; filling machine, enrobing machine; decorating device; folding unit, substrate feeding unit, manufacturing line
Wine drinks: wine drink GWC Spritz GWC Syringe, wine drink GWC Hugo GWC Hugo, wine drink MAMBO MAMBO, wine drink LiebKatz Favorite cat , wine drink SKIF LEGEND Legend of the Scythians .
Technological equipment for the food industry: Gear syringe-dispenser
Vacuum syringes: IPKS-047, IPKS-018 with spare parts
Vacuum syringe
Korzhik, a syringe with cottage cheese, a juicer with fruit filling, puff tongues, Belgian waffles
Technological equipment for the food industry: Piston syringe dispenser,
High-transparency polypropylene plastic packaging - disposable syringe 3 component 5.0 ml, type Luer.
Equisect® paste is a medicinal product for veterinary use in the form of an oral paste for the treatment and prevention of nematodes and gadfly infestations in horses. Packing: in polymer syringes-dispensers
assortment of oriental flour sweets: kurabye Yuzhnoye, kurabye Aigul, kurabye Baku, sand sticks, erebuni Vostochnye, horseshoe with jam , syringe with cottage cheese, juice with jam, Bakinsky nan, Baki nan
Cuts from beef and pork syringed.
Syringe dispenser D01 according to TU 9444-216-49640047-2015:
Sterile biopolymer prosthesis of synovial fluid in a disposable syringe for intra-articular administration according to TU 9398-002-00470622-2016 in the following versions:
Technological equipment for the food, meat and dairy and fish industries: vacuum syringes (stuffing pumps) of the IPKS-047 type
BIOLEK Tuberculin PPD-L, [solution for intradermal administration, 2 TE dose, 1 ml (10 doses of 2 TE in 0.1 ml) vials (1) + syringes (5) + needles for extraction (long) (5) + needles for injection (short) (5)] (1), car
Enoxaparin sodium solution for injection 8000 anti-Ha IU 0.8 ml, 0.8 ml syringes (10), cardboard packs
Automatic washing syringe installation, supply voltage 380 Volts,
Programmable infusion syringe pump-dispenser for long-term infusion of medicines and infusion solutions,
Delivery Syringe from Russia to worldwide
🇺🇿 Manufactures Syringe from Uzbekistan
Sterile injection syringe single use Luer 2 ml with needles, Sterile injection syringe single use Luer 5 ml with needles, Sterile injection syringe single application of Luer 10 ml with needles,
Decapeptyl Depot powder for preparation, suspension for injection 3.75 mg (syringes) complete with solvent 1 ml No. 1 (syringes) Decapeptil Solution for injection 0.1 mg / ml 1 ml No. 7 (syringe ampoules)
Single-use injection syringe, sterile with a 5 ml needle (0.7 x 32 mm), Single-use injection syringe, sterile with a 10 ml needle (0.7 x 38 mm )
syringe
Syringe
🇰🇬 TOP exporter Syringe from Kyrgyzstan
Infusion syringe pumps IP-12
infusion syringe pumps Aitecs2016
Get Current Price on Syringe
- Step 1: Contact sellers & inquire about Syringe
- Step 2: Receive quotes from the seller
- Step 3: Tell the seller to send you a trade assurance order contract
- Step 4: Accept the contract & make payment.
- Transaction level
- Buyer ratings & reviews
- Recent transactions
- Trade capacity
- Production capacity
- R&D capacity
We have large supplier & product base, Buyer safety measures, Best prices Logistics & quality inspection support, bulk Request for quote
| Export to city | Date | Km |
|---|---|---|
| Yekaterinburg (Russia) - Hong Kong (Hong Kong) | 07.2020 | 5764 |
| Mahilyow (Belarus) - Ulaanbaatar (Mongolia) | 07.2020 | 5147 |
| Bishkek (Kyrgyzstan) - Bishkek (Kyrgyzstan) | 07.2020 | 0 |
| Nizhniy Novgorod (Russia) - Ashgabat (Turkmenistan) | 07.2020 | 2306 |
| Novosibirsk (Russia) - Chisinau (Moldova) | 07.2020 | 3787 |
| Qaraghandy (Kazakhstan) - Tallinn (Estonia) | 07.2020 | 3206 |
| Andijon (Uzbekistan) - Temirtaū (Kazakhstan) | 07.2020 | 1033 |
| Farg‘ona (Uzbekistan) - Regensburg (Germany) | 07.2020 | 4687 |
| Osh (Kyrgyzstan) - Odense (Denmark) | 07.2020 | 4749 |
| Samarkand (Uzbekistan) - Albacete (Spain) | 07.2020 | 5766 |
| Vitsyebsk (Belarus) - Batumi (Georgia) | 07.2020 | 1727 |
| Hrodna (Belarus) - Khiwa (Uzbekistan) | 07.2020 | 3020 |
| Kara-Balta (Kyrgyzstan) - Piedras Negras (Mexico) | 07.2020 | 12043 |
| Tashkent (Uzbekistan) - Panevėžys (Lithuania) | 07.2020 | 3596 |
| Taraz (Kazakhstan) - Cayenne (French Guiana) | 07.2020 | 12264 |
| Homyel’ (Belarus) - Fredericton (Canada) | 07.2020 | 6575 |
| Namangan (Uzbekistan) - Hämeenlinna (Finland) | 07.2020 | 3856 |
| Tokmok (Kyrgyzstan) - Ventspils (Latvia) | 07.2020 | 4031 |
| Saint Petersburg (Russia) - Khorugh (Tajikistan) | 07.2020 | 3820 |
Where is Syringe made. Supplier map

 33
33 5
5